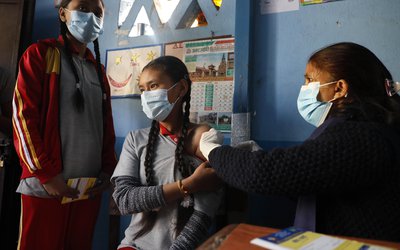
The world's first phase III clinical trial of the COVID-19 inactivated vaccine officially kicked off Tuesday. The ceremony of "Sinopharm's China National Biotec Group (CNBG) COVID-19 inactivated vaccine International Clinical (Phase III) trial and the United Arab Emirates" was held simultaneously in Beijing and Abu Dhabi via video conference.
CNBG and UAE Group 42 signed a clinical cooperation agreement. This is the first company in China to conduct an overseas clinical trial of the COVID-19 vaccine.
During the ceremony, the UAE Minister of Health & Prevention, Abdul Rahman Mohammed Al Owais, issued a Phase III of the COVID-19 inactivated vaccine clinical approval certificate to Ni Jian, Chinese Ambassador to the UAE. At the same time, Dr. Ali Obaid Al Dhaheri, UAE Ambassador to China, issued the Phase III clinical approval certificate of the COVID-19 inactivated vaccine to the Chair of CNBG Yang Xiaoming.
The ceremony held simultaneously in Beijing and Abu Dhabi via video conference. Experts say clinical research is usually divided into three phases.
Phase I mainly evaluates safety. Phase II evaluates mostly the safety and immunogenicity of vaccines and explores immunization procedures. Phase III mainly evaluates the safety and effectiveness of the vaccine in a larger population.
Safety evaluation runs through the entire clinical research process. If the test is successful, these vaccines are safe and effective and can protect vaccinated population from COVID-19.
Experts said that because the domestic epidemic was effectively controlled and did not meet the conditions for Phase III clinical trials, it actively promoted overseas cooperation in Phase III clinical trials.
This inactivated vaccine has passed phase I and II clinical trials, showing no "serious adverse reactions" with all volunteers generating antibodies after two doses in 28 days, its developer CNBG said.
The human trials began in Wuzhi County, central China's Henan Province, on April 12. A total of 1,120 volunteers aged 18-59 were inoculated with two doses in 28 days, according to CNBG. In the randomized, double-blind and placebo-controlled trial, all volunteers generated high levels of neutralizing antibodies, which can block pathogens from infecting human cells, the company said.

The study is the world's first clinical trial to obtain safety and effectiveness data of a two-dose inactivated COVID-19 vaccine, CNBG said, adding that the clinical trial has lasted the longest period of time (66 days) and got the most comprehensive data, as well as the most satisfying results among all COVID-19 vaccines to date.
On April 12, the clinical trial of the inactivated COVID-19 vaccine developed by its subsidiary Wuhan Institute of Biological Products got approved by the National Medical Products Administration, and the vaccine clinical trials started in China's Henan Province the same day.
In the past two months, the company has worked to build the world's largest, highly-efficient COVID-19 inactivated vaccine production facility, with construction standards and quality levels meeting international requirements. Experts say that the vaccine could enter the market by the end of 2020 or early next year. The facility is able to produce more than 200 million doses of inactivated COVID-19 vaccines annually.
China now has five COVID-19 vaccines approved for phase I clinical trials: one adenovirus vector vaccine and four inactivated vaccines, accounting for 40 percent of the total number of clinical trial stage vaccines worldwide, according to the Ministry of Science and Technology.
Source: CGTN
- Nepal-China Aid Project Meeting Held In Lhasa
- Apr 25, 2024
- Ambassador Subedi Presented The Letter Of Credence To President Of Italy
- Apr 25, 2024
- Nepal-Qatar Joint Business Council Formed
- Apr 25, 2024
- Weather Forecast: Isolated Brief Rain Is Likely To Occur At Few Places Of Koshi, Sudurpaschim And Karnali
- Apr 25, 2024
- Qatar And Nepal Ink Several MoUs, Returned Home Completing Two Days Visit
- Apr 24, 2024