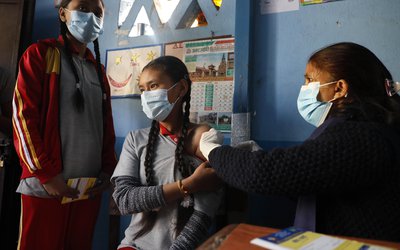
Bangladesh’s state medical research agency has approved a third-phase trial of a potential COVID-19vaccine developed by China’s Sinovac Biotech Ltd , as infections rise in the densely populated South Asian country.
Sinovac has been looking for volunteers outside China as the number of coronavirus cases there has dwindled, said a member of Bangladesh’s national technical advisory committee to tackle COVID-19.
The trial, to be conducted by the International Centre for Diarrhoeal Disease Research, Bangladesh (ICDDR,B), could begin next month.
“We have given ethical permission for the trial after reviewing the research protocol,” Mahmood Uz Jahan, director of the Bangladesh Medical Research Council (BMRC), told Reuters.
The trial will involve 4,200 volunteers, Jahan said.
“Half of them will get vaccinated,” he said.
The trial would be conducted in seven hospitals specialising in treating novel coronavirus infections in Dhaka, Bangladesh’s capital, an ICDDR,B official said on the condition of anonymity.
A Sinovac representative told Reuters that it was waiting for official approval from Bangladesh.
A senior Bangladesh health ministry official, who declined to be identified, said there should not be any objection from the government as Bangladesh would “get priority” if they can successfully develop the vaccine.
Bangladesh had 204,525 confirmed coronavirus cases as of Sunday, with 2,618 deaths. Sinovac said this month it was starting Phase III trials of its potential coronavirus vaccine in Brazil.
First human trial of AstraZeneca COVID-19 vaccine shows promise
AstraZeneca’s experimental COVID-19 vaccine was safe and produced an immune response in early-stage clinical trials in healthy volunteers, data showed on Monday, with the strongest response seen in people who received two doses. The vaccine, called AZD1222 and being developed by AstraZeneca and scientists at Britain’s University of Oxford, did not prompt any serious side effects and elicited antibody and T-cell immune responses, according to trial results published in The Lancet medical journal.
“There is still much work to be done before we can confirm if our vaccine will help manage the COVID-19 pandemic, but these early results hold promise,” vaccine developer Sarah Gilbert said.
“We still do not know how strong an immune response we need to provoke to effectively protect against SARS-CoV-2 infection.”
Gilbert said researchers needed to learn more about COVID-19 and continue late stage trials which have already commenced.
AstraZeneca shares spiked higher, but then gave up some gains, to last trade up 0.4% on the day.
AstraZeneca’s is among the leading vaccine candidates against a pandemic that has claimed more than 600,000 lives, alongside others in mid and late-stage trials.
These include shots being developed by China’s Sinovac Biotech, another from state-owned Chinese firm Sinopharm, and one from the U.S. biotech firm Moderna.
AstraZeneca has signed agreements with governments around the world to supply the vaccine should it prove effective and gain regulatory approval. The company has said it will not seek to profit from the vaccine during the pandemic.
Researchers said the vaccine caused minor side effects more frequently than a control group, but some of these could be reduced by taking paracetamol, with no serious adverse events from the vaccine.
AZD1222 was developed by Oxford university and licensed to AstraZeneca, which has put it into large-scale, late-stage trials to test its efficacy. It has also already signed deals to produce and supply over 2 billion doses of the shot.
The new trial included 1,077 healthy adults aged 18-55 years with no history of COVID-19.
“Today’s data increases our confidence that the vaccine will work and allows us to continue our plans to manufacture the vaccine at scale for broad and equitable access around the world,” said Mene Pangalos, Executive Vice President of BioPharmaceuticals Research and Development at AstraZeneca.
CanSino COVID-19 vaccine shows immune response in human trial
A COVID-19 vaccine developed by CanSino Biologics Inc (6185.HK) and China’s military research unit has shown to be safe and induced immune response in most of the recipients, researchers said on Monday.
The CanSino candidate, named Ad5-nCOV, is one of a handful of vaccines that have shown some promise in human testing and are gearing up for late-stage trials, along with projects involving Moderna Inc (MRNA.O), BioNTech SE (BNTX.O) and Inovio Pharmaceuticals Inc (INO.O).
While CanSino’s vaccine has yet to start late-stage large-scale clinical trials to assess how well it works to prevent people from becoming infected, it has received the greenlight to be used in China’s military.
The results from the mid-stage study, published in the medical journal Lancet, supports the testing of the vaccine candidate in a large trial, the study authors said.
The vaccine candidate uses a virus called adenovirus to carry genetic material from the new coronavirus’ protein into the human body, a method also used by researchers at the University of Oxford and AstraZeneca (AZN.L) for their vaccine candidate.
The immune responses elicited by the vaccine might be undermined if the inoculated person has already developed high-level adenovirus immunity from a previous infection, Chinese researchers said in a peer-reviewed paper.
CanSino counts Lilly Asia Ventures, backed by U.S. pharmaceutical giant Eli Lilly and Co (LLY.N), as its top shareholder, according to Refinitiv data.
- DigitALL: Innovation And technology For Gender Equality
- Mar 08, 2023
- India Says It Will Protect Its Interests As Chinese Boat Heads To Sri Lanka
- Jul 30, 2022
- Omicron Poses 'Very High' Global Risk, World Must Prepare: WHO
- Nov 29, 2021
- Resumption Of India's COVAX Vaccine Supply Delayed By Nepal
- Nov 24, 2021
- US Shipping 1.5 Million J&J Vaccines To Nepal
- Jul 10, 2021