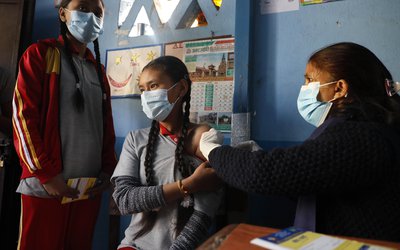
About 53 persons participating in the Covid-19 vaccine trials at the Post Graduate Institute of Medical Education and Research (PGIMER) have not reported any major side effects seven days after being vaccinated reports Hindustan Times.
Russia’s first vaccine, approved by the country’s regulator on August 12, had been widely criticised because it had not undergone phase-3 clinical trials, something that is being initiated only now.
The Indian Express reports that vaccine is already being administered in Russia, and is planned to be used by other countries as well, including India. Hyderabad-based Dr Reddy’s Laboratories has entered into an agreement to distribute the vaccine in India. But before it can be used on Indian population, the Russian vaccine, as is the case with any vaccine developed outside the country, would have to undergo late stage clinical trials in India. Those trials are expected to begin next month.
Russia has been developing another coronavirus vaccine, its second, and is speeding up its development to ensure an approval by the middle of this month, a report in The Wall Street Journal said.
Russia’s first vaccine, approved by the country’s regulator on August 12, had been widely criticised because it had not undergone phase-3 clinical trials, something that is being initiated only now. It was later clarified that the approval was only conditional, and for emergency use, and that it was contingent on phase-3 trials being conducted later.
The Wall Street Journal said the second vaccine was being developed by a former Soviet bioweapons research laboratory called Vector State Virology and Biotechnology Centre. It quoted the Russian Health Minister as saying that he expected the vaccine to get the approval as early as mid-October.
Hunt for coronavirus vaccine: The story so far
193 vaccine candidates in pre-clinical or clinical trials
42 of them in clinical trials
Ten in final stages, phase-III of human trials
At least eight candidate vaccines being developed in India. Two of these have entered phase -II trials after completing phase-I.
The ones most talked about:
* AstraZeneca/Oxford University
* Moderna
* Pfizer/BioNTech
* Johnson & Johnson
* Sanofi/GlaxoSmithKline
* Novavax
* Russian vaccine, developed by Gamaleya Institute in Moscow
* Three Chinese vaccines that have been approved for use in China without phase-3 trials being completed. One of them has been given emergency use authorisation in UAE
- Israel On Alert Against Terror Attacks, Airstrikes On Gaza Continue,
- Apr 23, 2024
- North Korea Says It Held Firing Drills Involving 'Super-large Rocket Launchers'
- Apr 23, 2024
- Russia Steps Up Security Measure In Moscow
- Apr 22, 2024
- Mount Everest Royalty: Over Rs 500 Million Collected in royalty
- Apr 21, 2024
- Finance Minister Discusses With US Government Officials And Officials Of The Millennium Challenge Corporation (MCC)
- Apr 21, 2024