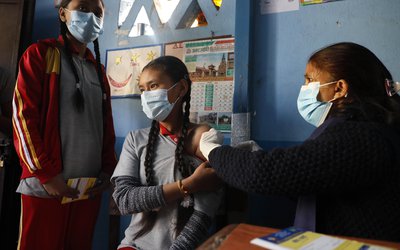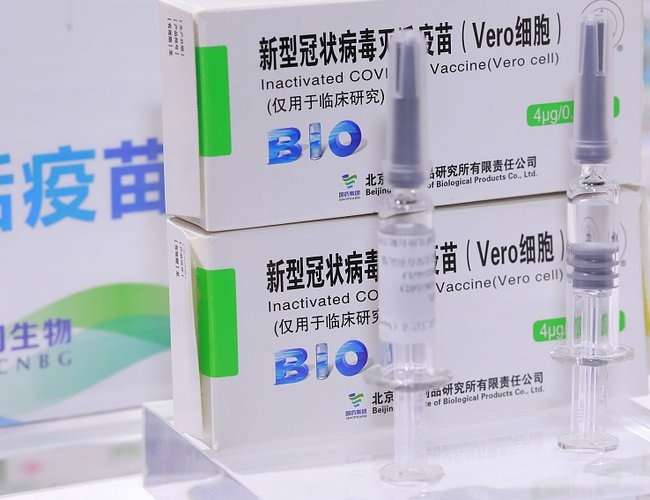
A Chinese COVID-19 vaccine candidate based on the inactivated whole SARS-CoV-2 virus is safe and elicits an antibody response, according to findings from a small early-phase randomized clinical trial published Thursday in The Lancet Infectious Diseases journal.
The latest study included participants aged between 18 and 80 years, and found that antibody responses were induced in all recipients. Participants aged 60 and older were slower to respond, taking 42 days before antibodies were detected in all recipients compared with 28 days for participants aged 18-59.
The trial was not designed to assess efficacy of the vaccine, so it is not possible to say whether the antibody responses induced by the vaccine, called BBIBP-CorV, are sufficient to protect people from SARS-CoV-2 infection.
BBIBP-CorV is developed by Beijing Institute of Biological Products, which is affiliated to state-owned China National Pharmaceutical Group (Sinopharm).
The authors also noted that interpretation of the results of this study is limited by the short duration of follow-up.
According to The Lancet, there are currently 42 vaccines for COVID-19 in clinical trials. Vaccine candidates in clinical trials vary in type and include DNA plasmid vaccines, inactivated virus vaccines, adenovirus-vectored vaccines, RNA vaccines, protein subunit vaccines and virus-like particle vaccines. Some of these have already proved safe and able to elicit immune responses in early phase clinical trials reports CGTN.
"Our findings indicate that a booster shot is necessary to achieve the greatest antibody responses against SARS-CoV-2 and could be important for protection. This provides useful information for a phase 3 trial," said Yang Xiaoming, one of the authors of the study and a professor at the Beijing Institute of Biological Products.
- Nepal Government Spends Only 52 Percent Of Targeted Annual Budget
- Apr 14, 2024
- Iran Launches Drones, Fires Missiles Toward Israel
- Apr 14, 2024
- Biden Urges Tehran Not To Proceed With Expected Attack On Israel
- Apr 13, 2024
- IAEA Warns Of Threats To Zaporizhzhia Nuclear Plant
- Apr 12, 2024
- Biden, Kishida Herald Partnekrship At Washington Summit
- Apr 11, 2024